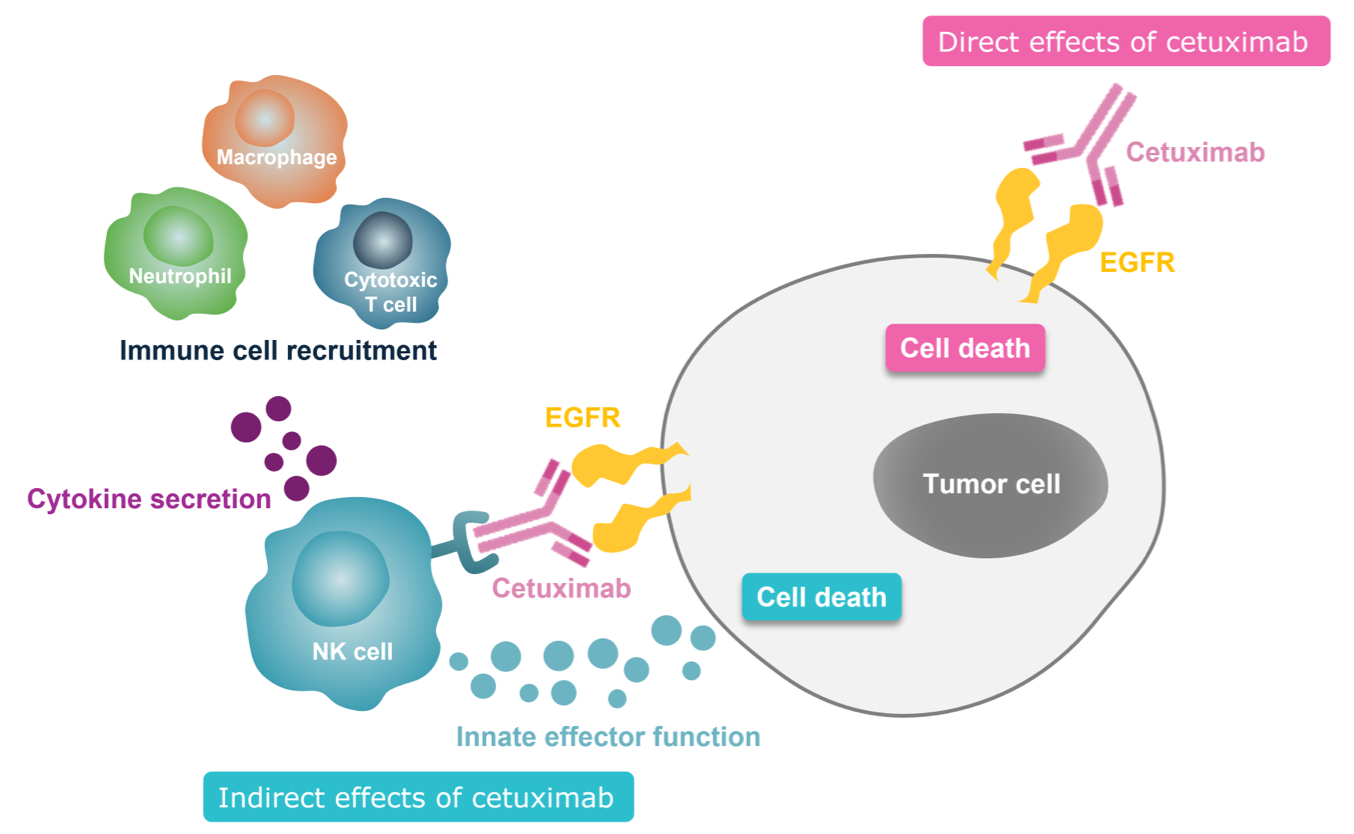
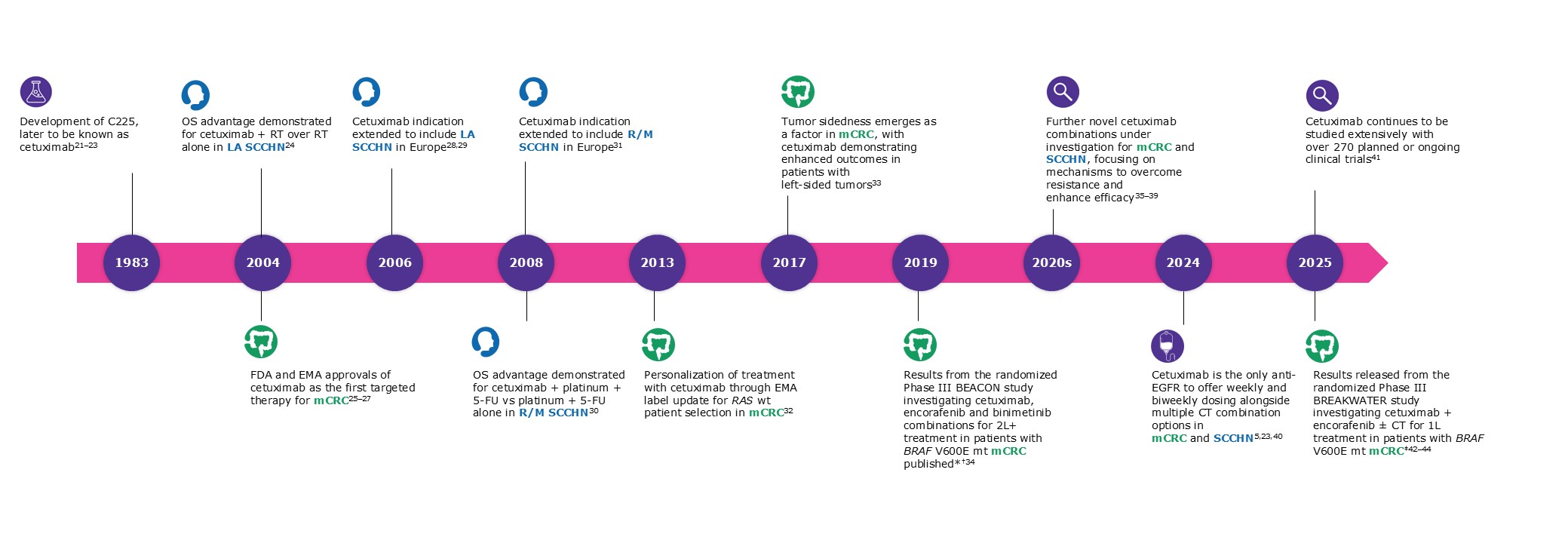
1L, first-line; ADCC, antibody-dependent cell-mediated cytotoxicity; CDC, complement-dependent cytotoxicity; CRC, colorectal cancer; CT, chemotherapy; EGFR, epidermal growth factor receptor; EMA, European Medicines Agency; FDA, Food and Drug Administration; HPV, human papillomavirus; ICD, immunogenic cell death; IgG, immunoglobulin G; LA, locally advanced; mCRC, metastatic colorectal cancer; MoA, mechanism of action; mt, mutant; NK, natural killer; OS, overall survival; R/M, recurrent and/or metastatic; RT, radiotherapy; SCCHN, squamous cell carcinoma of the head and/or neck; wt, wild-type.
 Global incidence and burden
Global incidence and burden Geographical variation
Geographical variation